Research Overview
Currently, we are living in an era when CO2 level in the atmosphere has become critical and our oceans are filling up with plastic waste. How can chemistry contribute in making our planet green again? To maintain sustainability, nature uses ‘catalysts’ to perform various chemical transformations, for example, photosynthesis, that is catalyzed by enzymes. In a related fashion, homogeneous catalysts can enable challenging chemical transformations under mild conditions and at the same time advance our mechanistic understanding at the molecular level. Embracing the principles of green chemistry and recently coined circular chemistry, we are interested in the development of new homogeneous catalysts and their applications in the service of clean energy and the environment.
Research Topics
Green Homogeneous Catalysis

We are interested in the development of green and sustainable catalytic processes for the waste-free production of useful organic compounds from feed stocks that are inexpensive or/and renewable. Our emphasis is on reactions that release or consume hydrogen gas (dehydrogenation or hydrogenation) as they have several advantages; for example, (a) (de)hydrogenation reactions are highly atom-economic and clean, (b) hydrogen gas can be produced from renewable sources for hydrogenation reactions and (c) for the dehydrogenation reactions, the produced hydrogen gas can be stored and used as a fuel. So, you can eat the cake and have it too! Moreover, we are also interested in developing efficient catalysts for the utilization of greenhouse gases such as CO2 and N2O for the synthesis of valuable chemicals.
New Hydrogen Storage Materials

Safe and long-term storage of hydrogen has been a longstanding hurdle in utilising hydrogen gas as a clean and renewable energy carrier. An attractive approach has been to use hydrogen-rich organic compounds, Liquid Organic Hydrogen Carrier (LOHC). LOHCs (charged fuel carriers) are small organic compounds in the liquid state at room temperature that can liberate hydrogen gas (fuel) in the presence of a catalyst forming spent fuel that can be again converted back to the charged fuel carrier, thus closing the loop. The liquid state gives the advantage of utilising the already established infrastructures of delivering gasoline fuels. The current target set for 2030 for the gravimetric capacity of chemical hydrogen storage materials (tank system level) by the European Union is 6.0 wt%. (De)hydrogenation of small organic molecules bearing high hydrogen storage capacity under mild conditions can be challenging which creates a strong need to develop efficient catalysts to tailor the purpose. We are interested in the development of efficient homogeneous catalysts for the utilization of small renewable organic compounds such as aqueous ethanol or glycerol as an LOHC.
Circular Chemistry
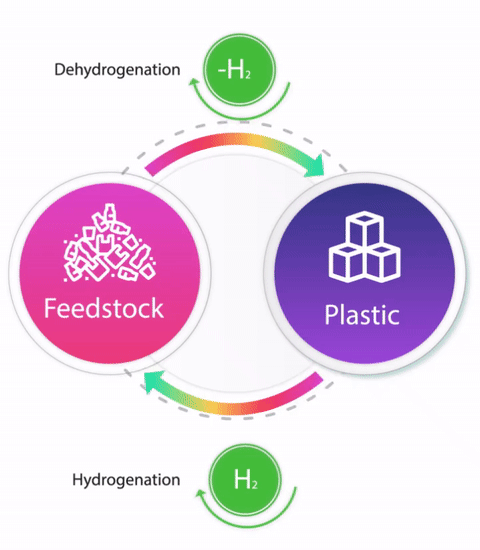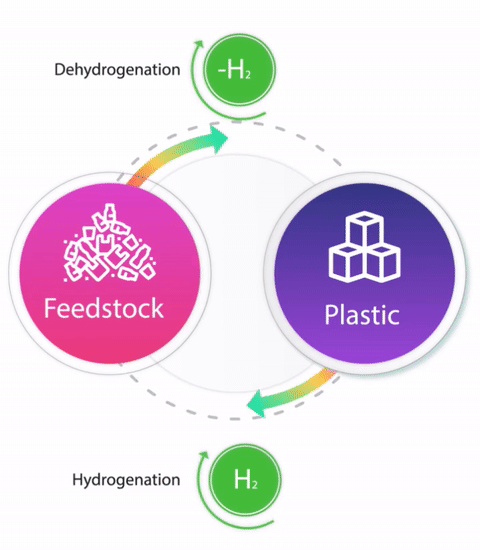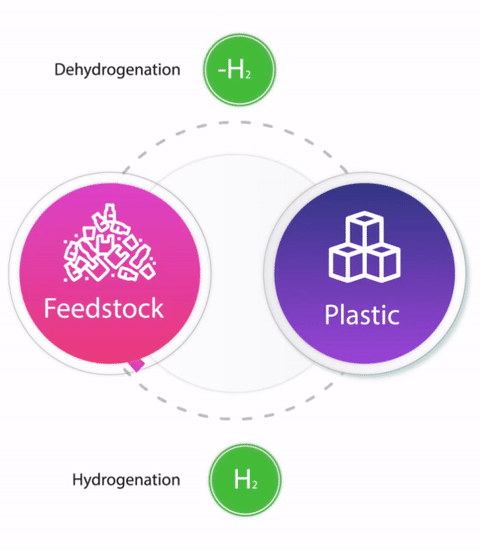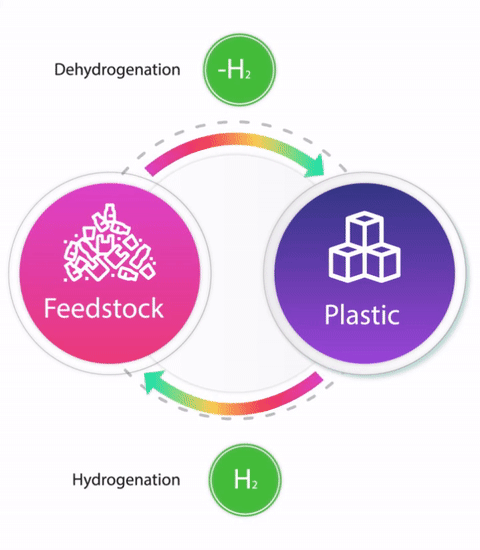
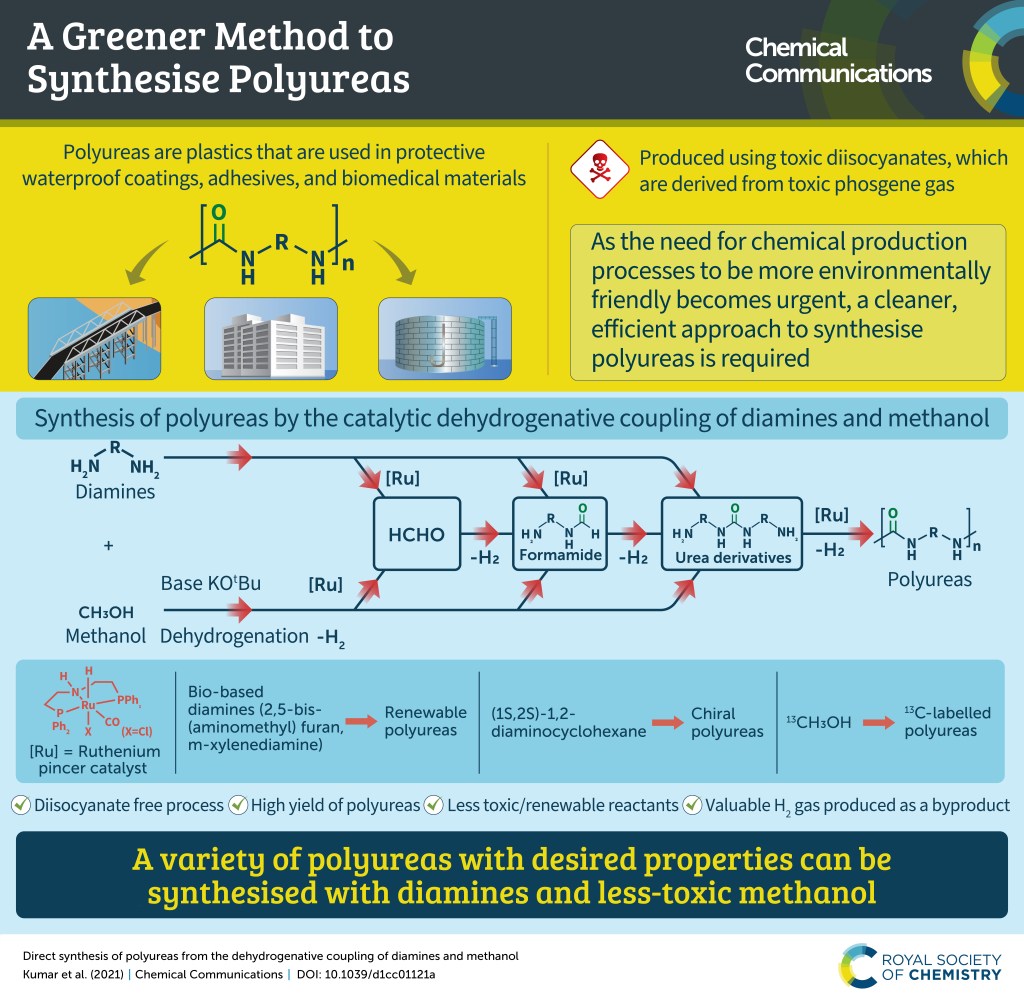
Linear industrial production where limited resources are continuously being used to produce valuable products which upon consumption is left to degrade is a root cause for several global crises such as climate change and shortage of energy and food. One of the approaches towards sustainability is Circular Economy which is defined as “restorative and regenerative by design, and aims to keep products, components, and materials at their highest utility and value at all times”. Chemistry being at the centre of industrial production plays an important role in achieving a circular economy. Circular chemistry takes into account all of the three, People, Planet, and Profit (‘Triple bottom line’) for one process and advocates that a process obeying all the principles of Green chemistry is not sustainable until it is economic. At the centre of all the principles of Circular Chemistry lies utilization of waste as a resource. We are interested in developing catalytic hydrogenation processes for the depolymerisation of plastic waste e.g. polyesters, polyamides, polycarbonates, polyurethanes, and polyureas to produce either monomers or other useful organic compounds. Moreover, we are interested in converting the depolymerised monomers/oligomers back to the fresh polymers using the reverse reaction i.e. catalytic dehydrogenation and closing the loop to demonstrate the circular economy of plastic production. For our recent discoveries, see: Chem. Commun., 2021, 57, 6153-6156; Eur. J. Org. Chem, 2021, 32, 4546; and ACS Catal, 2022, 12, 6923-6933.
Research Highlight